Efeitos da exposição aguda e crônica à diferentes salinidades da água em juvenis Piaractus brachypomus: Hematologia, bioquímica sanguínea, desempenho e histologia de fígado e brânquias
DOI:
https://doi.org/10.56926/repia.v5i1.147Palavras-chave:
espécie neotropical, fisiologia, crescimento vegetativo, choque osmóticoResumo
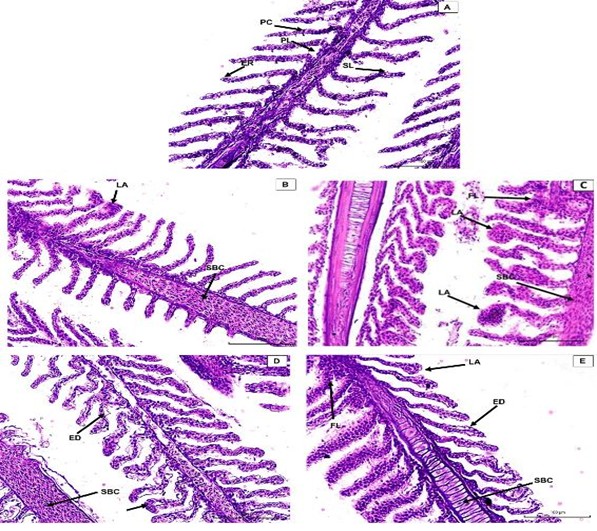
Esse estudo avaliou o efeito da exposição aguda e crônica a diferentes salinidades em juvenis de pirapitinga (Piaractus brachypomus). O experimento 1 avaliou a exposição aguda ao sal por 96 horas. O experimento 2 avaliou a exposição crônica a diferentes salinidades por 60 dias. Em ambos os experimentos foram identificadas alterações nos parâmetros sanguíneos e histológicos dos animais. No experimento 1, a salinidade de 12 g de sal L−1 se mostrou letal, com salinidades mais adequadas próximas a 3 g de sal L-1. No experimento 2, os melhores resultados de desempenho, após 60 dias, foram obtidos com até 2 g de sal L-1. Os maiores distúrbios nos parâmetros sanguíneos e as alterações teciduais mais intensas foram registrados para 4 e 6 g de sal L-1. Portanto, salinidade de até 2 g de sal L-1 se mostrou mais adequada para o cultivo de P. brachypomus.
Downloads
Referências
Abdel-Latif, H. M. R., Ahmed, H. A., Shukry, M., Khafaga, A. F., Elkhayat, B. K., Abdel-Tawwab, M., & Abd-elaziz, R. A. (2023). Growth performance, physiological responses, and histoarchitectural changes in juvenile Pangasianodon hypophthalmus under different environmental salinities. Fishes, 8(6), 282. https://doi.org/10.3390/fishes8060282
Abdel-Moneim, A. M., Al-Kahtani, M. A., & Elmenshawy, O. M. (2012). Histopathological biomarkers in gills and liver of Oreochromis niloticus from polluted wetland environments, Saudi Arabia. Chemosphere, 88(8), 1028–1035. https://doi.org/10.1016/j.chemosphere.2012.04.001
Abdel-Tawwab, M., Monier, M. N., Hoseinifar, S. H., & Faggio, C. (2019). Fish response to hypoxia stress: growth, physiological, and immunological biomarkers. Fish Physiology and Biochemistry, 45(3), 997–1013. https://doi.org/10.1007/s10695-019-00614-9
Amankwah, J. F., Jin, W., Ma, X., Xu, P., Wen, H., Amuneke, K. E., Munganga, B. P., Li, K., Liu, J., & Li, H. (2025). Salinity tolerance in freshwater Drum (Aplodinotus grunniens): Investigating biochemical, antioxidant, digestive enzyme, and gene expression responses to acute salinity stress. Animals, 15(7), 1015. https://doi.org/10.3390/ani15071015
Angeles‐Escobar, B. E., da Silva, S. M. B. C., & Severi, W. (2022). Growth, red blood cells, and gill alterations of red pacu (Piaractus brachypomus) fingerlings by chronic exposure to different total suspended solids in biofloc. Journal of the World Aquaculture Society, 53(3), 652–668. https://doi.org/10.1111/jwas.12837
Araújo, L. M. de, Gonçalves Junior, L. P., Silva, W. de S. e, & Luz, R. K. (2020). Salinity and prey concentration on larviculture of killifish Hypsolebias radiseriatus (Cyprinodontiformes: Rivulidae). Acta Scientiarum. Animal Sciences, 43, e52075. https://doi.org/10.4025/actascianimsci.v43i1.52075
Bao, J.-W., Qiang, J., Tao, Y.-F., Li, H.-X., He, J., Xu, P., & Chen, D.-J. (2018). Responses of blood biochemistry, fatty acid composition and expression of microRNAs to heat stress in genetically improved farmed tilapia (Oreochromis niloticus). Journal of Thermal Biology, 73, 91–97. https://doi.org/10.1016/j.jtherbio.2018.02.007
Blewett, T. A., Binning, S. A., Weinrauch, A. M., Ivy, C. M., Rossi, G. S., Borowiec, B. G., Lau, G. Y., Overduin, S. L., Aragao, I., & Norin, T. (2022). Physiological and behavioural strategies of aquatic animals living in fluctuating environments. Journal of Experimental Biology, 225(9). https://doi.org/10.1242/jeb.242503
Chen, X., Li, B., Hou, Y., Wei, K., Zhou, L., Zhang, C., Zhang, L., Zhu, J., & Jia, R. (2025). Physiological responses and serum metabolite alterations in Grass Carp (Ctenopharyngodon idellus) under chronic salinity exposure. Antioxidants, 14(11), 1287. https://doi.org/10.3390/antiox14111287
Dagoudo, M., Mutebi, E. T., Qiang, J., Tao, Y.-F., Zhu, H.-J., Ngoepe, T. K., & Xu, P. (2023). Effects of acute heat stress on haemato-biochemical parameters, oxidative resistance ability, and immune responses of hybrid yellow catfish (Pelteobagrus fulvidraco × P. vachelli) juveniles. Veterinary Research Communications, 47(3), 1217–1229. https://doi.org/10.1007/s11259-022-10062-1
Dagoudo, M., Qiang, J., Bao, J.-W., Tao, Y.-F., Zhu, H.-J., Tumukunde, E. M., Ngoepe, T. K., & Xu, P. (2021). Effects of acute hypoxia stress on hemato-biochemical parameters, oxidative resistance ability, and immune responses of hybrid yellow catfish (Pelteobagrus fulvidraco × P. vachelli) juveniles. Aquaculture International, 29(5), 2181–2196. https://doi.org/10.1007/s10499-021-00742-1
Dawood, M. A. O., Gewaily, M. S., & Sewilam, H. (2022). The growth performance, antioxidative capacity, and histological features of intestines, gills, and livers of Nile tilapia reared in different water salinities and fed menthol essential oil. Aquaculture, 554, 738122. https://doi.org/10.1016/j.aquaculture.2022.738122
de Morais Carvalho Ananias, I., dos Santos Silva, S., de Sena Souza, A., dos Santos, F. A. C., Magalhães, T. B., de Freitas Martins, W. J., Favero, G. C., & Luz, R. K. (2025). Effects of stocking densities on the growth performance and physiology of juvenile Piaractus brachypomus in recirculating aquaculture system. Fishes, 10(4), 171. https://doi.org/10.3390/fishes10040171
de Oliveira, C. G., Boaventura, T. P., Santos, F. A. C. dos, Souza, A. de S., Silva, S. dos S., Gil, C. S. B., Luz, R. K., & Oréfice, R. L. (2025). Effects of polypropylene microparticles ingestion on the growth and physiology of Piaractus brachypomus, and its accumulation and tissue damage. Aquatic Toxicology, 283, 107338. https://doi.org/10.1016/j.aquatox.2025.107338
Demska-Zakęś, K., Gomułka, P., Rożyński, M., & Zakęś, Z. (2021). Effect of a short-term sodium chloride bath on juvenile pikeperch (Sander lucioperca) welfare. Aquaculture Reports, 19, 100569. https://doi.org/10.1016/j.aqrep.2020.100569
do Carmo Neves, L., Favero, G. C., Beier, S. L., Ferreira, N. S., Palheta, G. D. A., de Melo, N. F. A. C., & Luz, R. K. (2020). Physiological and metabolic responses in juvenile Colossoma macropomum exposed to hypoxia. Fish Physiology and Biochemistry, 46(6), 2157–2167. https://doi.org/10.1007/s10695-020-00868-8
dos Santos Silva, S., Ananias, I. de M. C., Magalhaes, T. B., Martins, W. J. de F., Souza, A. de S., dos Santos, F. A. C., Melo, N., Murgas, L. D. S., Paschoalini, A. L., Bazzoli, N., Favero, G. C., & Luz, R. K. (2026). Effects of osmotic shock on survival, hematological and metabolic responses, antioxidant status and gill histology of juveniles of tambaqui (Colossoma macropomum) and the hybrid tambatinga (C. macropomum × Piaractus brachypomus). Aquaculture, 612, 743077. https://doi.org/10.1016/j.aquaculture.2025.743077
Escobar L., M. D., Ota, R. P., Machado‐Allison, A., Andrade‐López, J., Farias, I. P., & Hrbek, T. (2019). A new species of Piaractus (Characiformes: Serrasalmidae) from the Orinoco Basin with a redescription of Piaractus brachypomus. Journal of Fish Biology, 95(2), 411–427. https://doi.org/10.1111/jfb.13990
FAO. (2024). The State of World Fisheries and Aquaculture 2024. FAO. https://doi.org/10.4060/cd0683en
Ferreira, A. L., Bonifácio, C. T., de Souza e Silva, W., Takata, R., Favero, G. C., & Luz, R. K. (2021). Anesthesia with eugenol and menthol for Piaractus brachypomus (Cuvier, 1818): Induction and recovery times, ventilation frequency and hematological and biochemical responses. Aquaculture, 544, 737076. https://doi.org/10.1016/j.aquaculture.2021.737076
Ferreira, A. L., dos Santos, F. A. C., Bonifácio, C. T., & Luz, R. K. (2023). Effects of live prey concentration, salinity, and weaning age on larviculture of Piaractus brachypomus reared in a recirculating aquaculture system. Tropical Animal Health and Production, 55(2), 99. https://doi.org/10.1007/s11250-023-03514-6
Fiúza, L. S., Aragão, N. M., Ribeiro Junior, H. P., de Moraes, M. G., Rocha, Í. R. C. B., Lustosa Neto, A. D., de Sousa, R. R., Madrid, R. M. M., de Oliveira, E. G., & Costa, F. H. F. (2015). Effects of salinity on the growth, survival, haematological parameters and osmoregulation of tambaqui Colossoma macropomum juveniles. Aquaculture Research, 46, 1–9. https://doi.org/10.1111/are.12224
Griffith, M. B. (2016). Toxicological perspective on the osmoregulation and ionoregulation physiology of major ions by freshwater animals: Teleost fish, crustacea, aquatic insects, and Mollusca. Environmental Toxicology and Chemistry, 36(3), 576–600. https://doi.org/10.1002/etc.3676
Hoseini, S. M., Gupta, S. K., Yousefi, M., Kulikov, E. V., Drukovsky, S. G., Petrov, A. K., Taheri Mirghaed, A., Hoseinifar, S. H., & Van Doan, H. (2022). Mitigation of transportation stress in common carp, Cyprinus carpio, by dietary administration of turmeric. Aquaculture, 546, 737380. https://doi.org/10.1016/j.aquaculture.2021.737380
Hossain, F., Islam, S. M., Islam, M. S., & Shahjahan, M. (2022). Behavioral and histo-pathological indices of striped catfish (Pangasionodon hypophthalmus) exposed to different salinities. Aquaculture Reports, 23, 101038. https://doi.org/10.1016/j.aqrep.2022.101038
Huang, G., Li, H., Wang, J., Liao, T., Qiu, L., Xiong, G., Wang, L., Bai, C., & Zhang, Y. (2025). Pre-transport salt baths mitigate physiological stress and tissue damage in Channel Catfish (Ictalurus punctatus) fingerlings: Evidence from multi-biomarker assessment and histopathology. Animals, 15(15), 2249. https://doi.org/10.3390/ani15152249
Jahan, I., Chowdhury, G., Rafi, S., Ashab, M. A., Sarker, M., Chakraborty, A., Couetard, N., Kabir, M. A., Hossain, M. A., & Iqbal, M. M. (2024). Assessment of dietary polyvinylchloride, polypropylene and polyethylene terephthalate exposure in Nile tilapia, Oreochromis niloticus: Bioaccumulation, and effects on behaviour, growth, hematology and histology. Environmental Pollution, 345, 123548. https://doi.org/10.1016/j.envpol.2024.123548
Jomori, R. K., Luz, R. K., Takata, R., Fabregat, T. E. H. P., & Portella, M. C. (2013). Água levemente salinizada aumenta a eficiência da larvicultura de peixes neotropicais. Pesquisa Agropecuária Brasileira, 48(8), 809–815. https://doi.org/10.1590/S0100-204X2013000800001
Khor, L., Bodunde, O. A., Wills, R., Hanson, L., Adeyemo, O. K., Aina, O. O., Alarape, S. A., Delamare-Deboutteville, J., & Chadag, V. M. (2024). Understanding aquaculture biosecurity to improve catfish disease management in Ogun and Delta states, Nigeria. Aquaculture, 584, 740664. https://doi.org/10.1016/j.aquaculture.2024.740664
Liu, D., Zhang, Z., Song, Y., Yang, J., Lu, Y., Lai, W., Wu, Z., Zhao, D., Lin, H., Zhang, Y., Zhang, J., & Li, S. (2023). Effects of salinity on growth, physiology, biochemistry and gut microbiota of juvenile grass carp (Ctenopharyngodon idella). Aquatic Toxicology, 258, 106482. https://doi.org/10.1016/j.aquatox.2023.106482
Liu, Y., Tian, J., Song, H., Zhu, T., Lei, C., Du, J., & Li, S. (2025). Osmoregulation and physiological response of Largemouth Bass (Micropterus salmoides) juvenile to different salinity stresses. International Journal of Molecular Sciences, 26(8), 3847. https://doi.org/10.3390/ijms26083847
Luz, R. K., & Favero, G. C. (2024). Use of salt, anesthetics, and stocking density in transport of live fish: A review. Fishes, 9(7), 286. https://doi.org/10.3390/fishes9070286
Mattioli, C. C., Takata, R., Paes Leme, F. de O., Costa, D. C., Melillo Filho, R., de Souza e Silva, W., & Luz, R. K. (2017). The effects of acute and chronic exposure to water salinity on juveniles of the carnivorous freshwater catfish Lophiosilurus alexandri. Aquaculture, 481, 255–266. https://doi.org/10.1016/j.aquaculture.2017.08.016
Mazumder, S. K., Das, S. K., Bakar, Y., & Ghaffar, M. A. (2016). Effects of temperature and diet on length-weight relationship and condition factor of the juvenile Malabar blood snapper (Lutjanus malabaricus Bloch & Schneider, 1801). Journal of Zhejiang University-SCIENCE B, 17(8), 580–590. https://doi.org/10.1631/jzus.B1500251
Mohamed, N. A., Saad, M. F., Shukry, M., El-Keredy, A. M. S., Nasif, O., Van Doan, H., & Dawood, M. A. O. (2021). Physiological and ion changes of Nile tilapia (Oreochromis niloticus) under the effect of salinity stress. Aquaculture Reports, 19, 100567. https://doi.org/10.1016/j.aqrep.2020.100567
Monier, M. N., Shady, S. H. H., Grana, Y. S., El-Ghaffar, H. A. A., Samir, F., El-Werwary, S. O. M., Ahmed, A. A., & Abdel-Tawwab, M. (2025). Interactive impact of salinity and oxygen level on the growth performance, digestive enzymes, serum biochemistry, antioxidative, immunity, and histological status of Nile tilapia (Oreochromis niloticus). Fish Physiology and Biochemistry, 51(6), 200. https://doi.org/10.1007/s10695-025-01608-6
Motamedi-Tehrani, J., Peyghan, R., Shahriari, A., Razijalali, M., & Ebrahimi, E. (2025). The influence of ammonia-N and salinity levels on oxidative stress markers, hepatic enzymes, and acid phosphatase activity in Nile tilapia (Oreochromis niloticus). Scientific Reports, 15(1), 559. https://doi.org/10.1038/s41598-024-84136-2
Nakadate, K., Sono, C., Mita, H., Itakura, Y., & Kawakami, K. (2023). Severe acute liver dysfunction induces delayed hepatocyte swelling and cytoplasmic vacuolization, and delayed cortical neuronal cell death. International Journal of Molecular Sciences, 24(8), 7351. https://doi.org/10.3390/ijms24087351
Okomoda, V. T., Isah, S., Solomon, S. G., & Ikhwanuddin, M. (2024). Salinity tolerance in Clarias gariepinus (Burchell, 1822): insight on blood parameter variations and gill histological changes. Fish Physiology and Biochemistry, 50(2), 605–616. https://doi.org/10.1007/s10695-023-01293-3
Oladosu, G. A., & Oladosu, O. O. (2019). Antibiotic sensitivity and sodium chloride susceptibility patterns of Flavobacterium columnare isolated from clinical columnaris in cultured Clarias gariepinus. Journal of Veterinary Medicine and Animal Health, 11(3), 59–63. https://doi.org/10.5897/JVMAH2018.0724
Perry, S. F. (1998). Relationships between branchial chloride cells and gas transfer in freshwater fish. In Biochem. Physiol (Vol. 119, Number 1).
Prathib, P. S. (2025). The Red-Bellied Pacu: A South American fish making waves in Indian aquaculture.
Riaz, M. N., Hafeez‐ur‐Rehman, M., Azmat, H., & Mehmood, S. (2025). Salinity‐induced variations in growth performance, stress biomarkers and biochemical profiles of striped catfish ( Pangasianodon hypophthalmus, Sauvage, 1878). Journal of Fish Biology, 107(6), 2046–2055. https://doi.org/10.1111/jfb.70211
Seshagiri, B., Kumar, A., Pradhan, P. K., Sood, N., Kumar, U., Satyavati, C., Sravanti, Y., Prasoon, K., Ghosh, A., Kantharajan, G., Basheer, V. S., Chakrabarti, P. P., Das, P. C., Lal, K. K., & Jena, J. K. (2022). Farming practices and farmers’ perspective of a non-native fish red-bellied Pacu, Piaractus brachypomus (Cuvier, 1818) in India. Aquaculture, 547, 737483. https://doi.org/10.1016/j.aquaculture.2021.737483
Souza-Bastos, L. R., Bastos, L. P., Carneiro, P. C. F., & Freire, C. A. (2016). Acute salt exposure of the freshwater Characiformes: Pacu (Piaractus mesopotamicus, Holmberg 1887), Tambaqui (Colossoma macropomum, Cuvier 1818), and their hybrid “Tambacu” Aquaculture, 465, 352–358. https://doi.org/10.1016/j.aquaculture.2016.09.028
Souza, R. J. F., dos Santos Silva, S., da Silva, N. C. S., de Morais Carvalho Ananias, I., dos Santos, F. A. C., de Sena Sousa, A., Magalhães, T. B., de Oliveira, C. G., de Sales, S. C. M., & Luz, R. K. (2025). Hematological and blood biochemical responses of Colossoma macropomum and Piaractus brachypomus subjected to sudden temperature reduction and thermal readjustment. Fish Physiology and Biochemistry, 51(3), 87. https://doi.org/10.1007/s10695-025-01499-7
Strzyzewska, E., Szarek, J., & Babinska, I. (2016). Morphologic evaluation of the gills as a tool in the diagnostics of pathological conditions in fish and pollution in the aquatic environment: a review. Veterinární Medicína, 61(3), 123–132. https://doi.org/10.17221/8763-VETMED
Takata, R., Mattioli, C. C., Bazzoli, N., Júnior, J. D. C., & Luz, R. K. (2021). The effects of salinity on growth, gill tissue and muscle cellularity in Lophiosilurus alexandri juvenile, a Neotropical freshwater catfish. Aquaculture Research, 52(9), 4064–4075. https://doi.org/10.1111/are.15244
Tavares‐Dias, M. (2022). Toxicity, physiological, histopathological, handling, growth and antiparasitic effects of the sodium chloride (salt) in the freshwater fish aquaculture. Aquaculture Research, 53(3), 715–734. https://doi.org/10.1111/are.15616
Uehara, S. A., Walter, E. H. M., Cunha, D. M., Calixto, F. A. A., Furtado, A. A. L., Takata, R., & Mesquita, E. de F. M. (2021). Effects of salinity gradients on the skin and water microbial community and physiological responses of tilapia (Oreochromis niloticus) in the pre‐slaughter. Aquaculture Research, 52(10), 4638–4647. https://doi.org/10.1111/are.15298
Usman, S., Abdull Razis, A. F., Shaari, K., Amal, M. N. A., Saad, M. Z., Mat Isa, N., & Nazarudin, M. F. (2021). Polystyrene microplastics exposure: An insight into multiple organ histological alterations, oxidative stress and neurotoxicity in Javanese Medaka Fish (Oryzias javanicus Bleeker, 1854). International Journal of Environmental Research and Public Health, 18(18), 9449. https://doi.org/10.3390/ijerph18189449
Wells, R. M. G., & Pankhurst, N. W. (1999). Evaluation of simple instruments for the measurement of blood glucose and lactate, and plasma protein as stress indicators in fish. Journal of the World Aquaculture Society, 30(2), 276–284. https://doi.org/10.1111/j.1749-7345.1999.tb00876.x
Xing, G., Zhang, K., Gao, S., Hao, Y., Nie, Z., Wei, J., Ai, T., Zhang, S., Zhang, J., & Huang, Z. (2025). Structural adaptations to saline stress: Histomorphological changes in the osmoregulatory and metabolic organs of Perca schrenkii under acute and chronic challenges. Biology, 14(12), 1775. https://doi.org/10.3390/biology14121775
Yousefi, M., Hoseini, S. M., Weber, R. A., da Silva, E., Rajabiesterabadi, H., Arghideh, M., & Delavar, F. H. (2022). Alleviation of transportation-induced stress in Nile tilapia, Oreochromis niloticus, using brackish water. Aquaculture Reports, 27, 101378. https://doi.org/10.1016/j.aqrep.2022.101378
Zhu, Z., Li, S., Lei, C., Zhu, T., Tian, J., Du, J., Wei, S., & Song, H. (2025). Survival and acute osmoregulatory response of grass carp under salinity stress. Comparative Biochemistry and Physiology Part A: Molecular & Integrative Physiology, 308, 111905. https://doi.org/10.1016/j.cbpa.2025.111905
Zidan, E. M., Goma, A. A., Tohamy, H. G., Soliman, M. M., & Shukry, M. (2022). Insight study on the impact of different salinity levels on behavioural responses, biochemical stress parameters and growth performance of African catfish (Clarias gariepinus). Aquaculture Research, 53(7), 2750–2759. https://doi.org/10.1111/are.15790
Downloads
Publicado
Como Citar
Edição
Seção
Licença
Copyright (c) 2026 André de Sena Souza, Fabio Aremil Costa Santos, Gustavo Soares da Costa Júlio, Sidney dos Santos Silva, Imaculadade Morais Carvalho Ananias, Camila Gomes de Oliveira, Pedro Paulo Cortezzi Pedras, Gisele Cristina Favero, Ronald Kennedy Luz

Este trabalho está licenciado sob uma licença Creative Commons Attribution 4.0 International License.
Os autores mantêm os seus direitos:
a. Os autores conservam os direitos de propriedade intelectual (copyright) dos trabalhos publicados, cedendo à revista o direito de primeira publicação.
b. Os autores conservam os seus direitos de marca registada e de patente, bem como de qualquer processo ou procedimento descrito no artigo.
c. Os autores conservam o direito de partilhar, copiar, distribuir, executar e comunicar publicamente o artigo publicado na REPIA (por exemplo, colocando-o num repositório institucional ou publicando-o num livro), com um reconhecimento da sua publicação inicial na REPIA.
d. Os autores conservam o direito de publicar posteriormente o seu trabalho, de utilizar o artigo ou qualquer parte dele (por exemplo, uma compilação do seu trabalho, notas de conferência, tese ou para um livro), desde que indiquem a fonte de publicação (autores do artigo, revista, volume, número e data).
Traduzido com a versão gratuita do tradutor - DeepL.com







